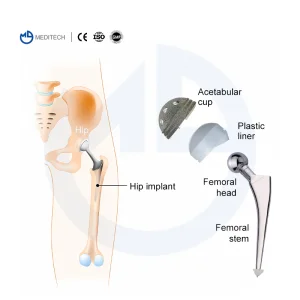
 Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement
Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement
 Surgical Instruments Posterior Cervical Instrument Set Hospital Used
Surgical Instruments Posterior Cervical Instrument Set Hospital Used
 PPSU Al All Kind of Instrument Box
PPSU Al All Kind of Instrument Box
 CE Certified Orthopedic Expert Femoral Instrument Set for Shoulder Surgery
CE Certified Orthopedic Expert Femoral Instrument Set for Shoulder Surgery
 Orthopedic Toolmed Knee Prosthesis Manufacturer Knee Replacement Arthroplasty Tka
Orthopedic Toolmed Knee Prosthesis Manufacturer Knee Replacement Arthroplasty Tka
 Prosthesis Replacement Orthopedic Sterilized Package Class III Knee Joint
Prosthesis Replacement Orthopedic Sterilized Package Class III Knee Joint
 Replacement Sterilized Package Prosthesis Knee Joint 3D Printed
Replacement Sterilized Package Prosthesis Knee Joint 3D Printed
 S-ROM Titanium Alloy Cementless Hip Joints for Revision Arthroplasty Surgery
S-ROM Titanium Alloy Cementless Hip Joints for Revision Arthroplasty Surgery
The global orthopedic revision market is entering a phase of unprecedented growth and technological sophistication. As primary total hip arthroplasty (THA) and total knee arthroplasty (TKA) procedures have scaled globally over the last three decades, the natural "lifecycle" of these implants has led to an increasing volume of revision surgeries. This is a critical sector of the medical device industry, catering to patients who require replacement of their original implants due to aseptic loosening, periprosthetic infection, mechanical failure, or osteolysis.
Industrial data indicates that the Revision Hip and Knee market is projected to expand at a CAGR of over 5.5% through 2030. In regions like North America and Europe, the demand is driven by an aging population that remains physically active, while in emerging markets across the Asia-Pacific and Latin America, the volume is surging due to improved healthcare access and the maturation of early primary procedures. For Revision Hip/Knee Systems Manufacturers & Suppliers, the challenge lies in creating modular, adaptable systems that can handle significant bone loss and compromised soft tissue environments.
Leading manufacturers are now shifting from "one-size-fits-all" components to modular systems that allow surgeons to reconstruct joint geometry with millimeter precision. The industrial landscape is no longer just about forging metal; it is about biological integration, 3D lattice structures for osseointegration, and instrument sets that simplify the complex logistics of revision surgery.

Our flagship S-ROM Titanium Alloy Cementless Hip Joints represent the pinnacle of revision technology. Specifically engineered for patients with complex femoral geometry, these systems offer a modular stem design that decouples proximal and distal sizing. This allows for optimal stability in the diaphysis while maintaining anatomical alignment in the proximal femur, essential for successful revision outcomes.
The use of biocompatible titanium alloy ensures long-term survival, while the textured surface promotes rapid secondary fixation. As a leading manufacturer, we ensure that every component is tested for fatigue resistance, simulating decades of human movement before it ever leaves our facility.
Additive manufacturing is revolutionizing revision surgery. Custom 3D-printed titanium augments and cages are now used to manage massive acetabular defects, providing a level of bone-ingrowth potential that traditional casting cannot match.
AI-driven surgical planning is becoming the standard. Our systems are designed to integrate seamlessly with digital templating software, allowing surgeons to map out bone resection and implant placement with high accuracy.
Moving beyond basic alloys, the trend is toward highly cross-linked polyethylene (HXLPE) and ceramic-on-ceramic surfaces to minimize wear debris, the primary cause of osteolysis in primary failures.
For international hospital groups and medical device distributors, the procurement of Revision Hip/Knee Systems is a high-stakes decision. The criteria have moved beyond simple pricing to include supply chain resilience, sterilization reliability, and surgical support logistics. We understand that a hospital in Germany has different regulatory and clinical workflow requirements than a clinic in Southeast Asia.
Our localized application scenarios include:
As a global supplier, we provide not just the hardware, but the complete surgical ecosystem—from the Disposable Pulsed Lavage System for site preparation to the PPSU Instrument Boxes that withstand repeated autoclave cycles.
Innovation meets biology with our 3D Printed Knee Joint Prosthesis. By utilizing advanced laser-sintering technology, we create a trabecular metal structure that mimics the architecture of natural bone. This is particularly crucial in knee revision, where the tibial platform must find stability on often compromised bone beds.
The sterilized packaging ensures that the implant arrives in the operating room in pristine condition, ready for immediate use. This system significantly reduces the need for bone cement in certain clinical scenarios, fostering a more biological long-term fix.

Chengdu Medev Medical Instruments Co., Ltd. was founded in 2001, covering an area of 18,000 ㎡, including a floor area of over 15,000 ㎡. Our registered capital reaches 20 million Yuan, and we specialize in research, design, and manufacturing of orthopedic implant instruments. As a leader in the industry, we have supplied 1000+ clients in 120+ countries for over 20 years.

Cleaned implants have a cleaner surface, helping to reduce the risk of post-operative infection. Ensures that the implant is safer to apply to the patient during surgery.

Tests the tensile strength, yield strength and elongation of materials. Simulates the tensile forces to which the implant is subjected in the body.

Packaging ensures a good seal to prevent entry of contaminants. We use heat sealing and pressing to ensure the integrity of the sterile barrier.
Choosing a Chinese factory like Chengdu Medev for your Revision Hip/Knee Systems provides a unique competitive edge in today’s volatile market. Our advantages are built on three pillars: Technical Precision, Scalable Efficiency, and Rigorous Quality Control.
Firstly, the industrial clusters in China have matured, allowing us to access the highest grade of titanium and cobalt-chrome alloys at optimized costs. We pass these efficiencies directly to our clients without compromising on the strict ISO and CE standards required for Class III medical devices. Our 3D printing capabilities are at the forefront of the industry, allowing for rapid prototyping of custom revision components that would take months elsewhere.
Secondly, our R&D team collaborates with top surgeons to refine instrument sets. A common frustration in revision surgery is the "clutter" of too many instrument trays. We design our Expert Femoral Instrument Sets and Acetabular Reamer Sets to be intuitive, reducing surgical errors and OR time. This commitment to "the surgeon’s experience" is why we have maintained 20+ years of growth.
Finally, we adhere to the ‘people-oriented, integrity first, continuous innovation, the pursuit of excellence’ concept. Guarding human health is not just a slogan; it is reflected in our 100% inspection policy for every orthopedic implant that leaves our factory.
 Disposable Pulsed Lavage System
Disposable Pulsed Lavage System
 Medical Equipment Hip Joint Instrument Set (cut) Surgical Instrument Acetabular
Medical Equipment Hip Joint Instrument Set (cut) Surgical Instrument Acetabular
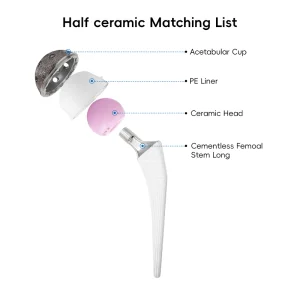 Orthopedic Prosthesis Implant Artificial Hip and Knee Joint Replacement
Orthopedic Prosthesis Implant Artificial Hip and Knee Joint Replacement
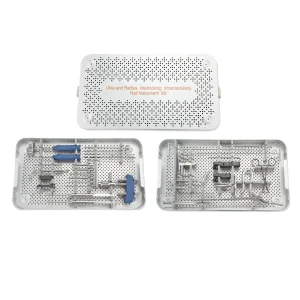 Orthopedic Medical Device Surgical Ulna and Radius Interlocking Instrument Set
Orthopedic Medical Device Surgical Ulna and Radius Interlocking Instrument Set
 Orthopedic Replacement Sterilized Package Prosthesis Knee Joint
Orthopedic Replacement Sterilized Package Prosthesis Knee Joint
 Locking Compression Plate (LCP) for Femur - LC-LCP Orthopedic Implant System
Locking Compression Plate (LCP) for Femur - LC-LCP Orthopedic Implant System
 Advanced Hip & Knee Prosthesis with Durable Materials
Advanced Hip & Knee Prosthesis with Durable Materials
 Replacement CE Sterilized, Carton Package Prosthesis Knee Joint
Replacement CE Sterilized, Carton Package Prosthesis Knee Joint