












| Still deciding? Get samples of $ ! US$ 1600/Set Request Sample |
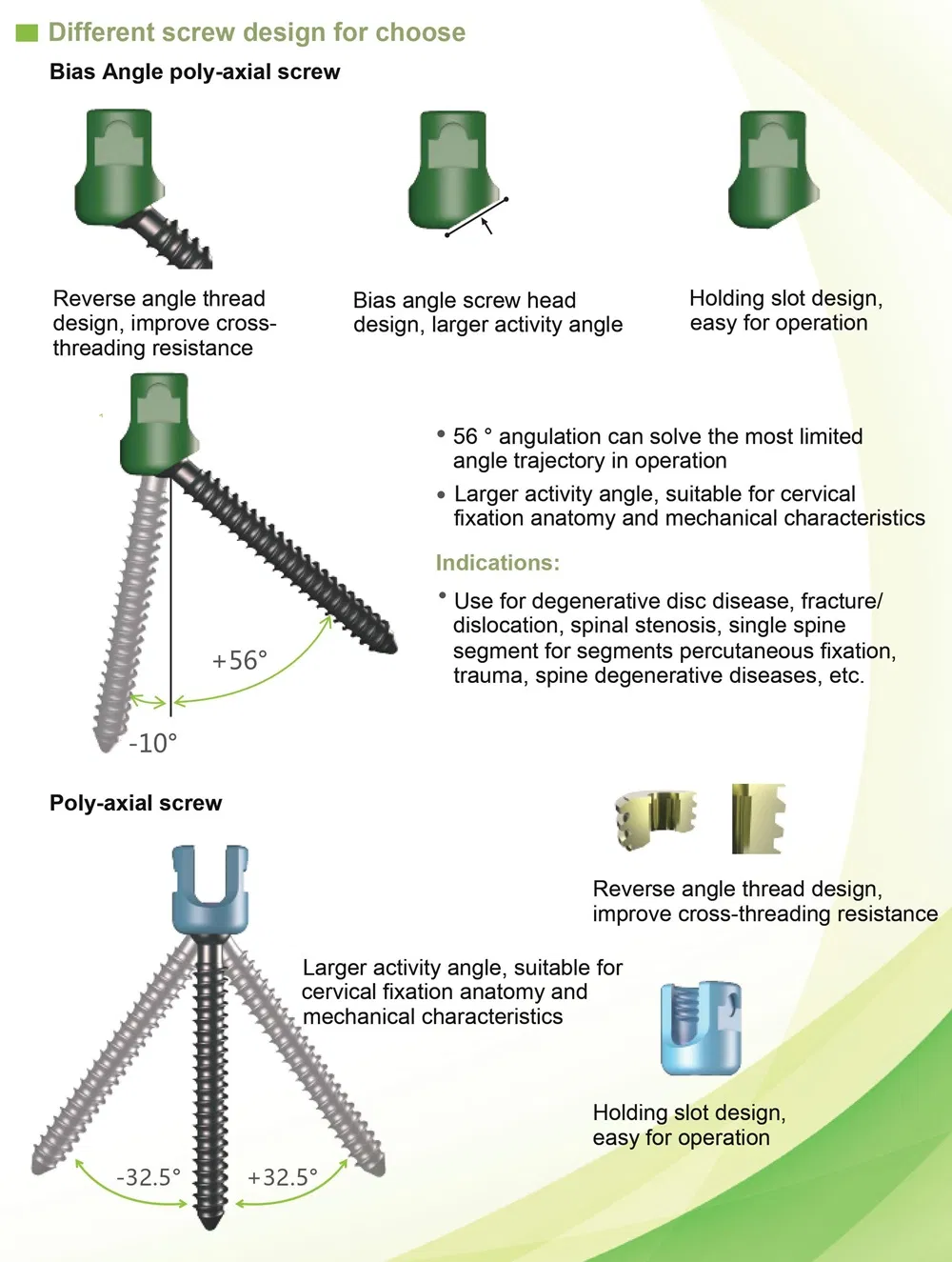
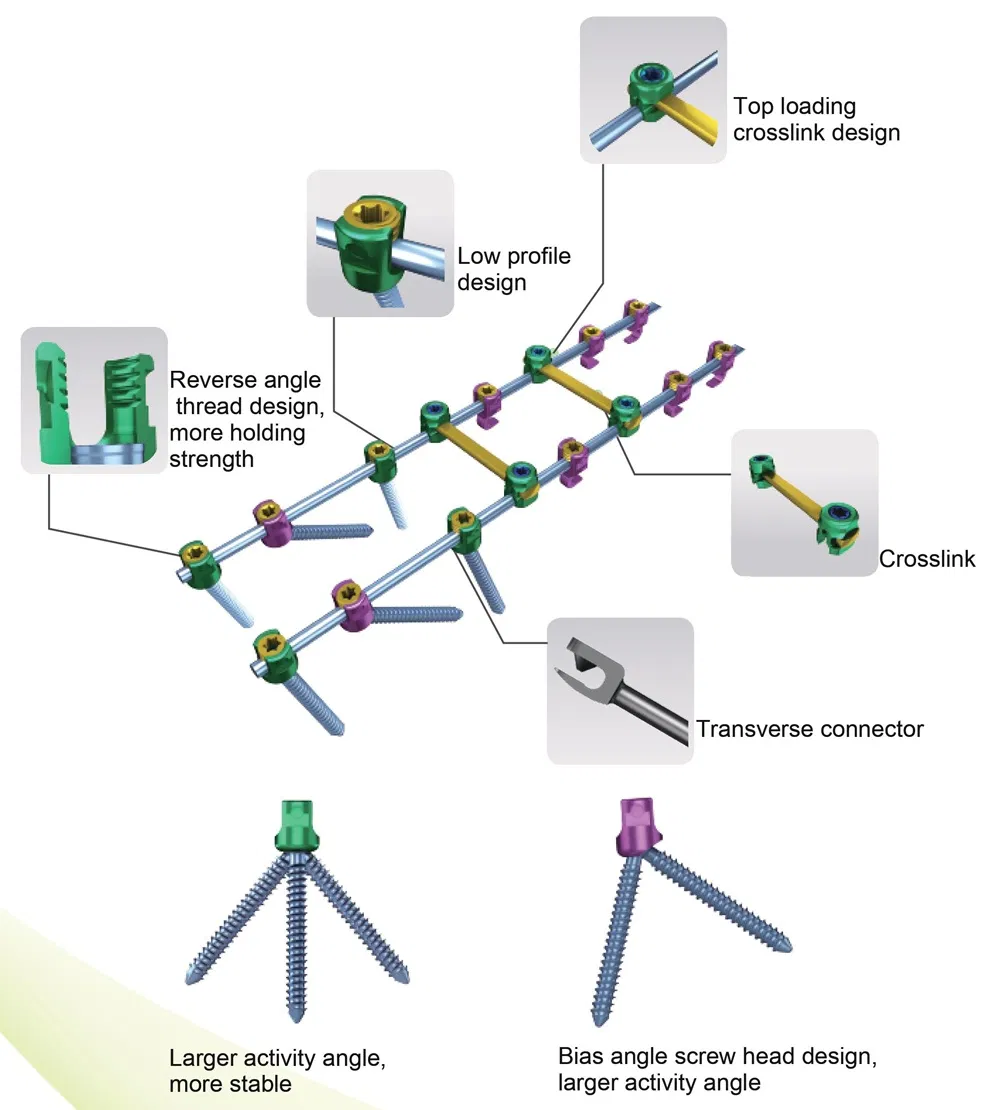
Key Features:
Clinical Applications:
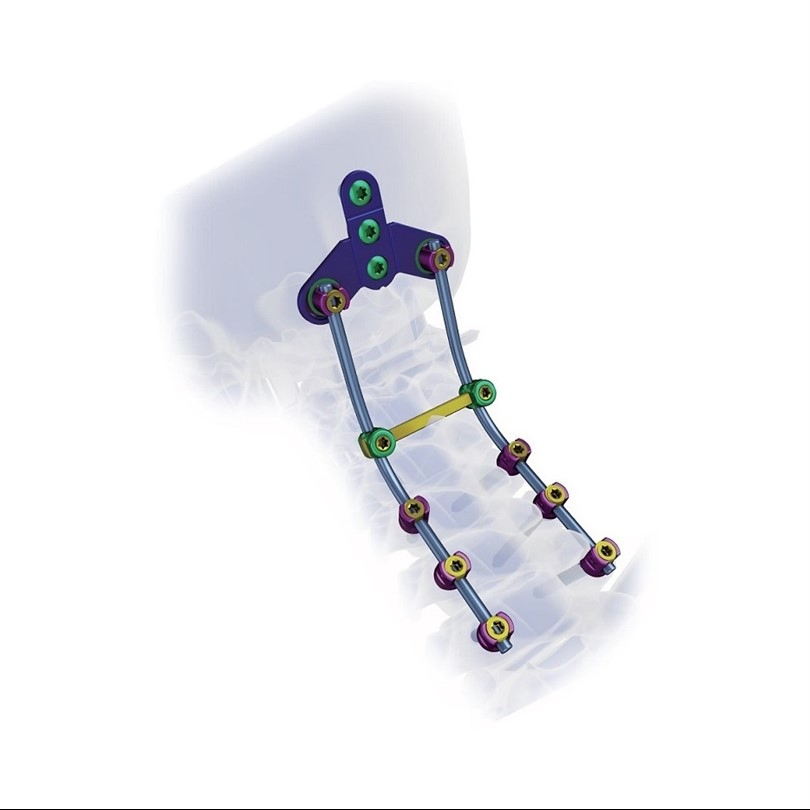
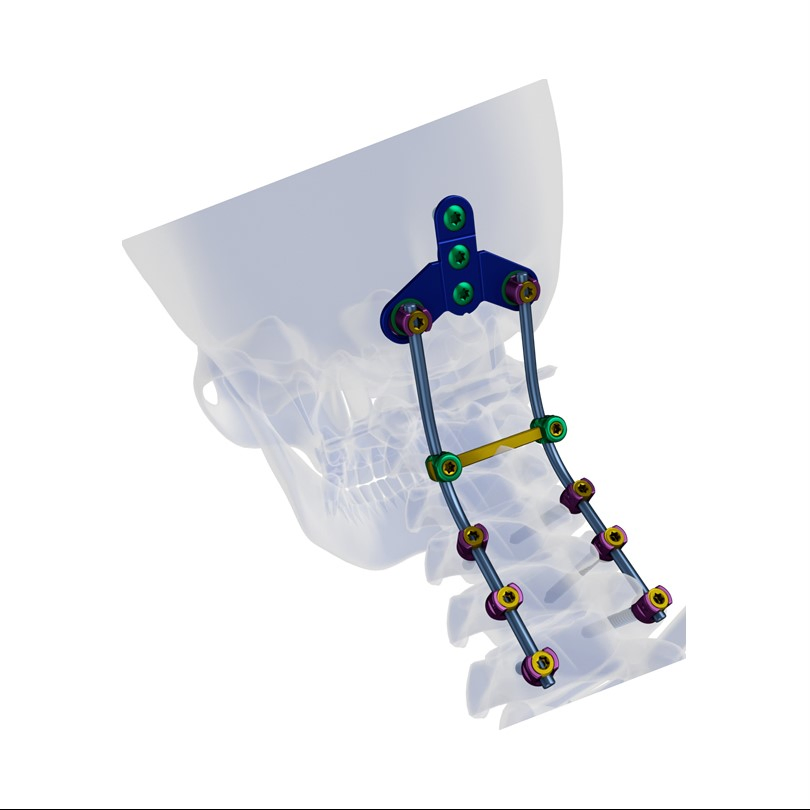
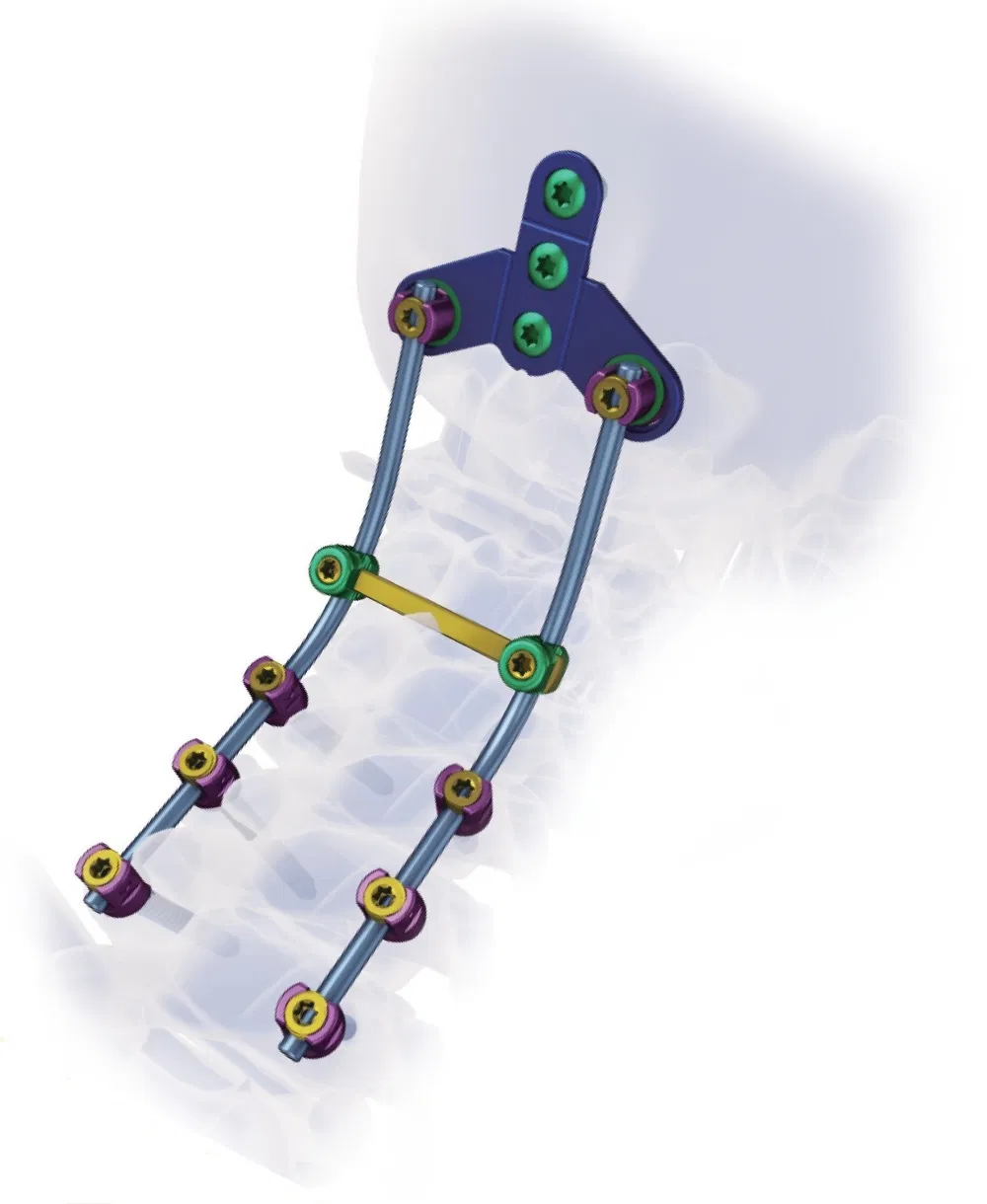
Excellent biocompatibility, ideal for treating degenerative disc diseases, fractures, dislocations, spinal stenosis, and occipital-cervical disassociation.


■ Packing Details:
- Implants: Inner plastic bag + outer carton box. Screws are packed in sets of 5; plates are in individual waterproof bags.
- Instruments Set: Professional instrument box + outer carton box.
- Customized packing designs are available upon request.
■ Delivery Way:
Reliable shipping via air and international couriers: DHL, UPS, FedEx, and more.

One-stop Orthopedic Solutions: Providing comprehensive orthopedic products since 2016, including spinal surgery series, metallic bone plates, screws, and intramedullary nails.
Production Capacity: Reaching 10,000 units per month, serving clients across Southeast Asia, the Middle East, Latin America, Africa, and Europe.

Factory Advantages:


 Medev Medical
Medev Medical