




1 / 5










| Still deciding? Get samples of $ ! US$ 88/Piece Request Sample |
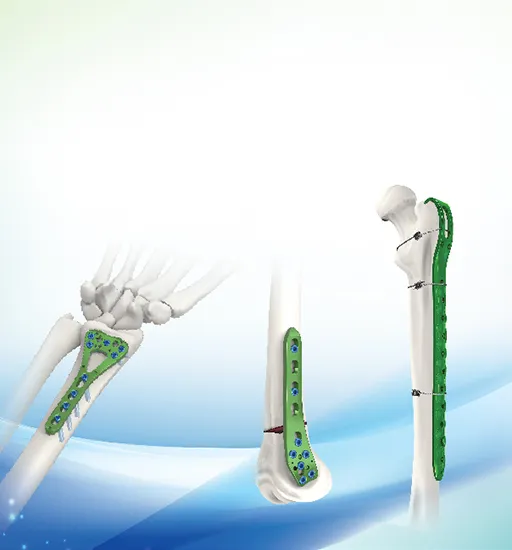
| Multi-axial Distal Femur Lateral Locking Plate 341 | ||
|---|---|---|
| P/N | Material | Holes |
| 34104-34113 L/R | Titanium | 4,5,6,7,8,9,10,11,12,13 holes |
| Compatible with 5.0 locking screw, 5.0 cortical self tapping screw | ||










 Medev Medical
Medev Medical