




1 / 5










| Still deciding? Get samples of $ ! US$ 18/Piece Request Sample |
| P/N | Product Name | Material | Model/Specifications |
|---|---|---|---|
| 18660-186115 | 7.3 Cannulated half thread locking screw | Titanium Alloy | 60-115mm (5mm size gap) |

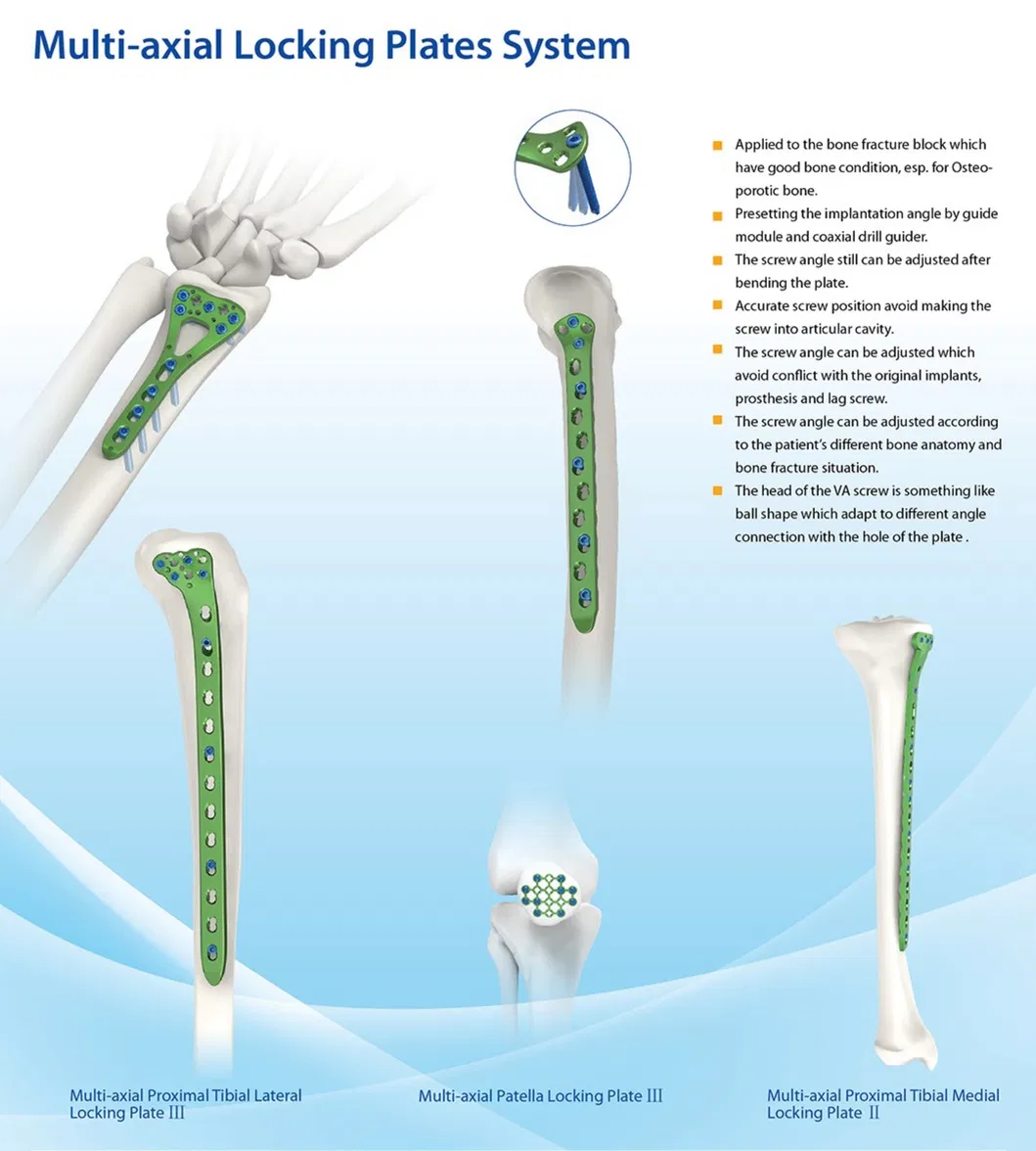
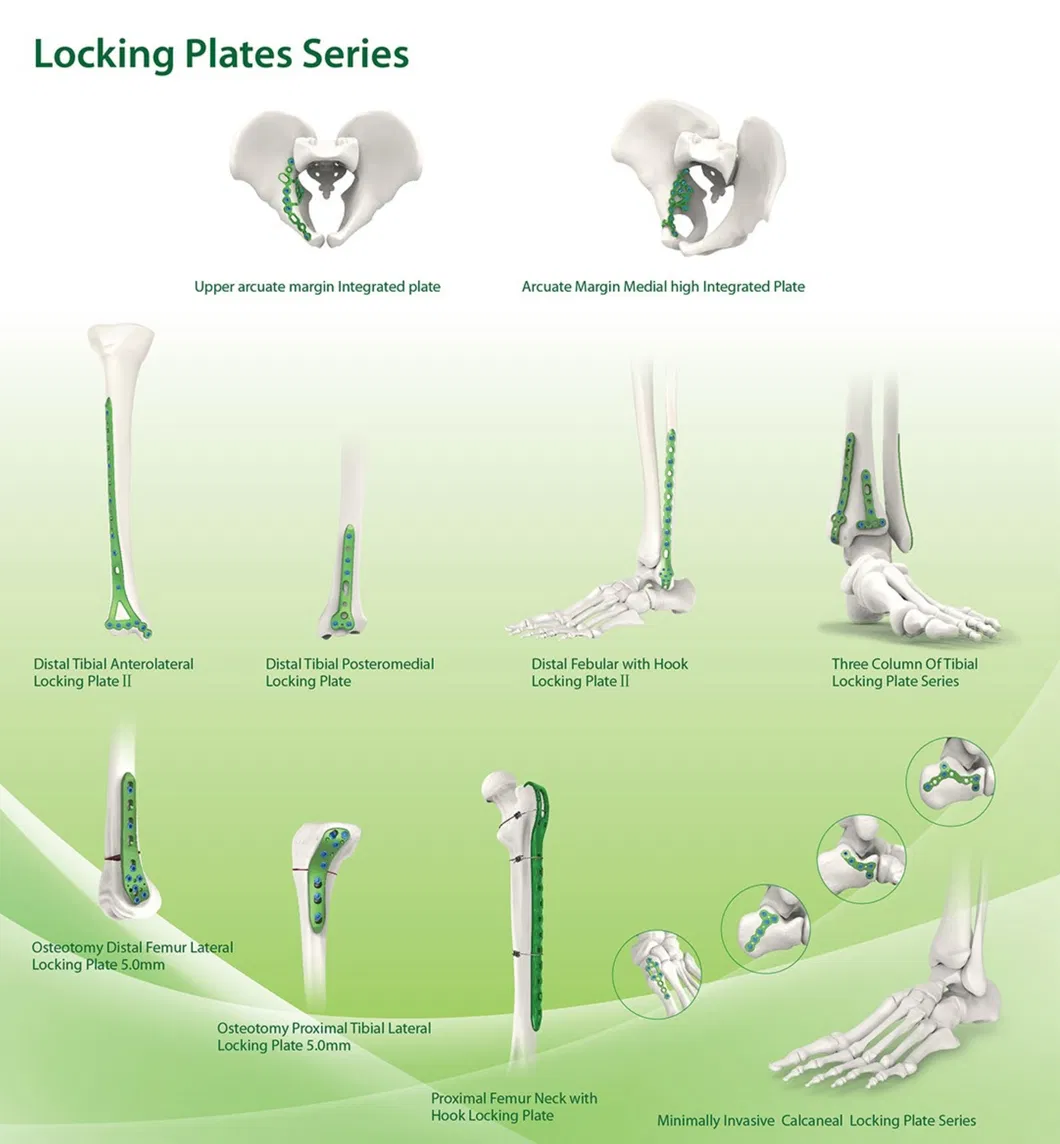

Established in 1958, we are engaged in the development, manufacture, and sales of orthopedic implants and instruments. As a key enterprise in the medical apparatus industry, our product line covers spine pedicle screws, interlocking nails, locking plates/screws, trauma plates, cannulated screws, external fixators, hip prosthesis, and surgical instruments.
All medical apparatus have passed national and international approvals. Our products are characterized by high-quality materials and innovative designs. We utilize advanced equipment, including imported CNC machines and high-precision digital control systems, within 100,000 Grade purification chambers to ensure the highest standards.



 Medev Medical
Medev Medical