Product Description
Disposable Peek Suture Anchor Orthopedic Implant Bone
| Product Name |
Non-bioabsorbable Suture Anchor |
| Carton Size |
42*22.5*25.5CM |
| G.W./CTN |
1.5KG-1.8KG |
| Certificates |
CE & ISO13485 |
| Validity Period |
3 Years |
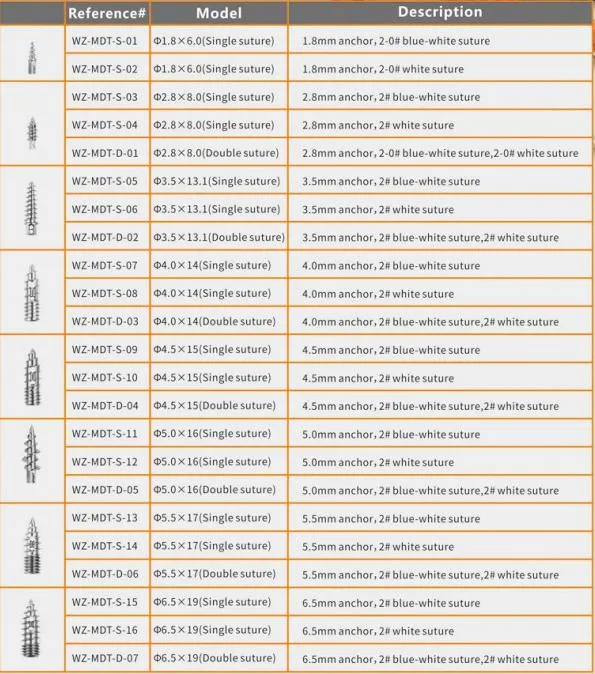
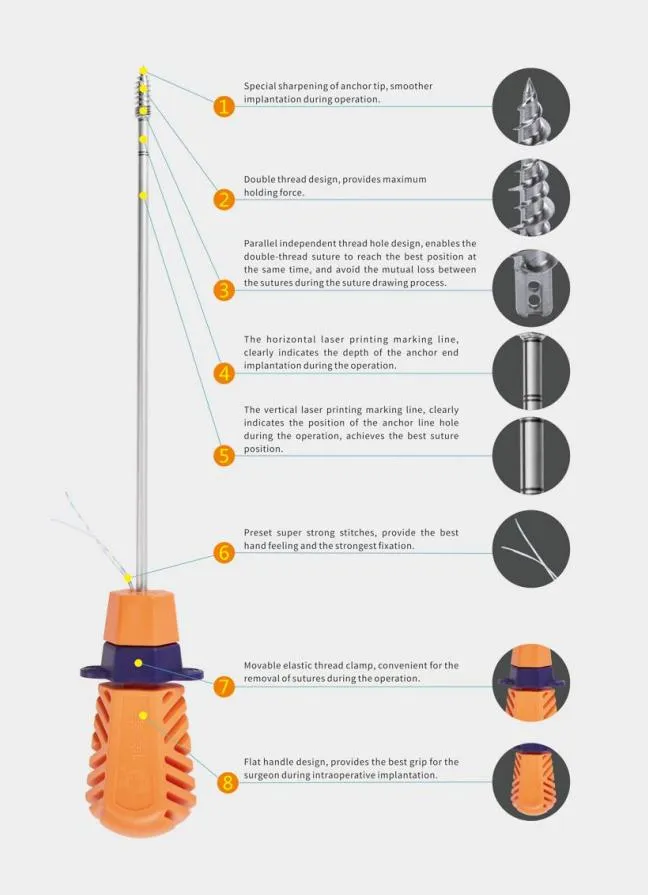
The Suture Anchor System consists of an anchor, nonabsorbable suture, and inserter. The anchor is constructed from Ti6Al4V alloy, conforming to ISO5832-3:2016 standards. The suture is crafted from ultra-high molecular weight polyethylene (UHMWPE) material without coating, woven from multiple UHMWPE yarns meeting ASTM F2848-17 requirements. The blue suture is dyed with FDA-approved blue material. Inserter parts contacting the human body are made of stainless steel (ASTM F899-12b), while the handle is made of polycarbonate and ABS material.
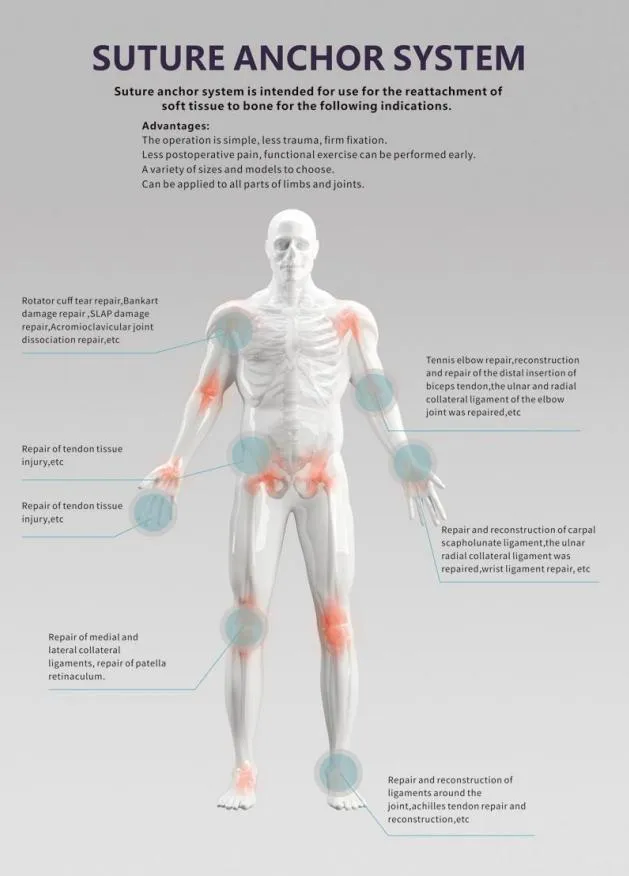
Surgical Application
Our system is designed for effective debridement following orthopedic procedures of trauma and joint arthroplasty. It combines convenience and power for the removal of contaminated water, necrotic tissue, bone debris, and foreign materials while providing controlled pressure for efficient irrigation on flat wound surfaces or intra-medullary sites.
Frequently Asked Questions
What are the main materials used in the anchor?
The anchor is made of high-quality Ti6Al4V alloy, which meets the rigorous requirements of ISO5832-3:2016 for orthopedic implants.
What certifications does this product hold?
The product is fully certified with CE and ISO13485, ensuring it meets international medical standards for safety and quality.
How long is the sterile shelf life?
The product has a period of validity of 3 years from the date of sterilization, provided the packaging remains intact.
Can I customize the logo on the products?
Yes, we support customization via laser marking. Please contact our service team for specific details and requirements.
What is the typical lead time for an order?
For ready-to-ship items, it takes approximately 1 workday. For new production, the lead time typically ranges from 5 to 30 workdays depending on the order volume.
What are the payment methods available?
We accept payments via TT (Telegraphic Transfer), B/L, and other standard international commercial payment terms.




















 Medev Medical
Medev Medical