














| Customization: | Available |
|---|---|
| Certification: | ISO, CE |
| Disinfection: | Disinfection |
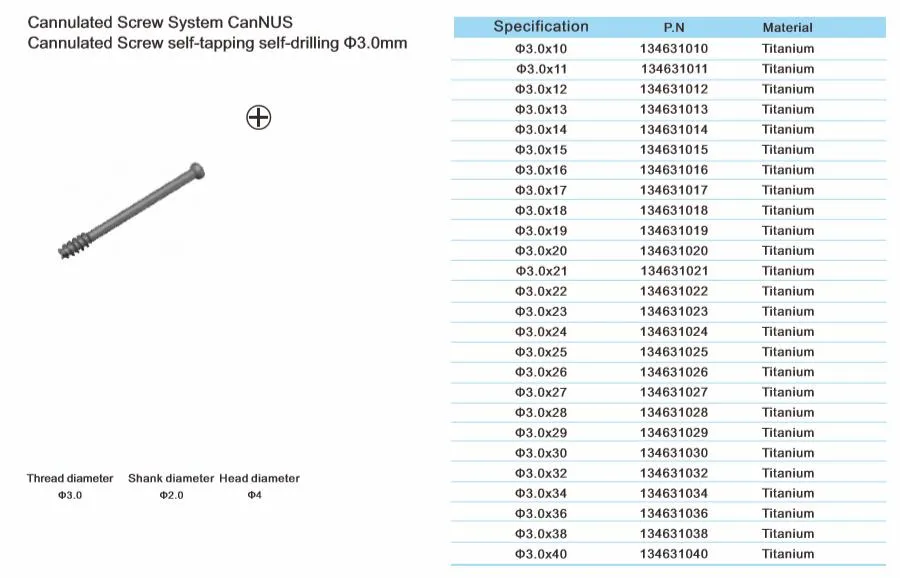
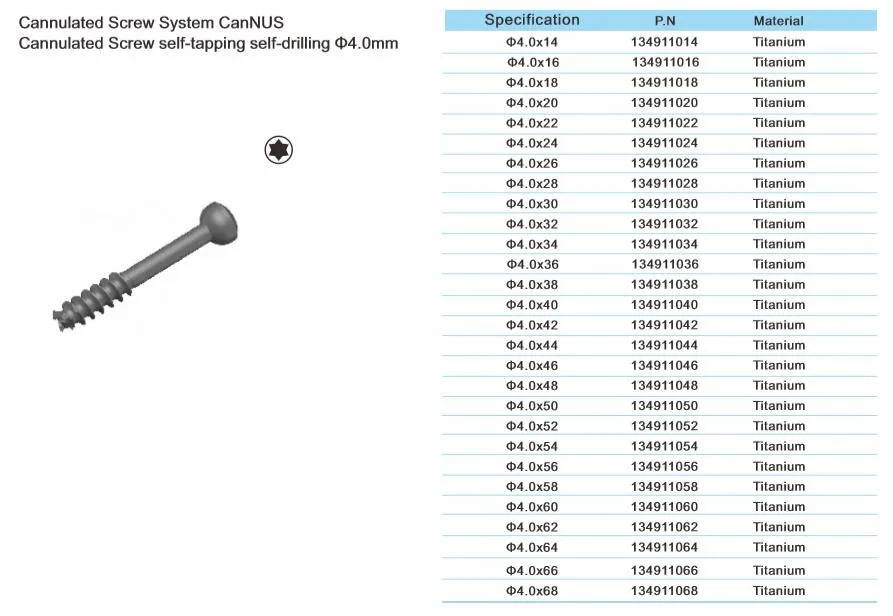
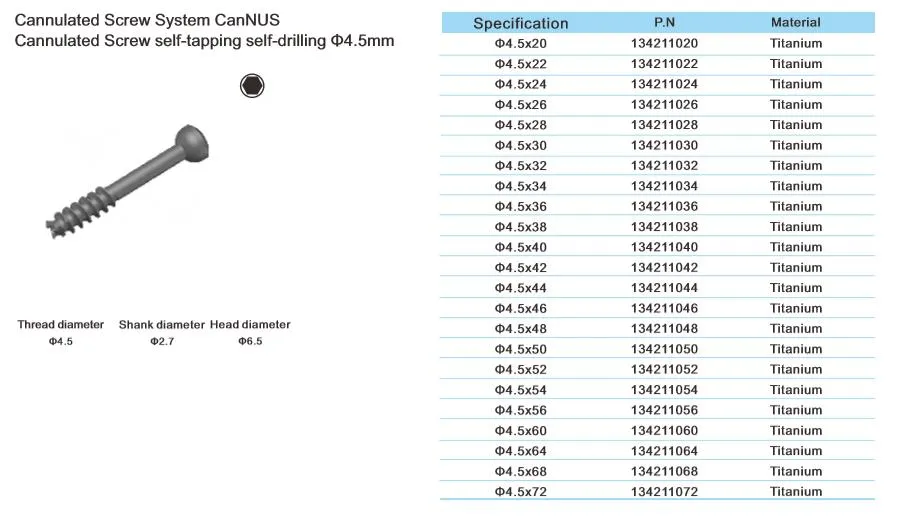
Cannulated Screw System CanNUS
Tornillos canulados
Cannulated Screw Φ6.5mm, self-drilling, thread length 32mm


A medical device company is one of the largest designers, manufacturers, and suppliers of orthopedic implants and instruments through 20 years' development. The product lines include traumatology, spinal fixation system and surgical instrument, covering products such as femoral nail, titanium plates and screws, pedicle screw with rods, PEEK cage, balloon kyphoplasty for spine fracture and other instrument sets.
Significant investment has been made to upgrade modern production equipment, improve production processes and develop superior technical skills. The goal is to provide the best orthopedics products and improve life quality for people in need.
A complete Quality Assurance System has been established to continuously fulfill the requirements according to ISO9001, 13485, QSR820, CE, ranging from material procurement to design development and automated production processes.
QMS consists of five subsystems:
Effectiveness of quality management system is continually improved to provide products and services which consistently meet and exceed expectations.
 Medev Medical
Medev Medical