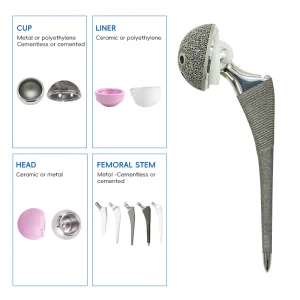
The global orthopedic surgery market is witnessing an unprecedented surge, with the acetabular cup segment positioned as a critical cornerstone. As a primary component in Total Hip Arthroplasty (THA), acetabular cups are essential for restoring mobility to millions of patients suffering from osteoarthritis, rheumatoid arthritis, and femoral neck fractures. Current market analysis indicates that the demand for high-quality orthopedic implants is growing at a CAGR of over 5%, driven largely by an aging global population and the increasing prevalence of obesity-related joint issues.
In the industrial sphere, the manufacturing of acetabular cups has transitioned from traditional forging to advanced 3D titanium printing and precision CNC machining. As a trusted Acetabular Cups Supplier, we recognize that surgeons today demand implants that offer not just longevity, but biological compatibility and "press-fit" stability. The shift towards "Cementless" fixation has revolutionized how we design the outer shell of the cup, utilizing porous surfaces that encourage natural bone ingrowth.
Years Experience
Countries Exported
Global Clients
Production Base
Ultra-High Molecular Weight Polyethylene (UHMWPE) remains the gold standard for acetabular liners, offering superior wear resistance and durability under heavy physiological loads.
Application of Hydroxyapatite (HA) and Titanium plasma spray coatings enhances osseointegration, ensuring a permanent bond between the implant and the pelvis.
Utilizing AI-driven quality control and digital twin simulations to predict implant behavior within the human body, reducing the risk of post-operative failure.
Our UHMWPE Insert Acetabular Cup is engineered for maximum biocompatibility. It serves as the bearing surface in total hip replacements, significantly reducing friction against the femoral head. Designed for both Hemiarthroplasty and Total Hip Replacement, this system provides the flexibility surgeons need for diverse patient anatomies.
Procurement departments in international hospitals and medical device distributors are increasingly looking for cost-effective yet high-compliance solutions. In developed markets like North America and Europe, the focus is on "revision surgery" solutions and highly cross-linked polyethylene. Conversely, in emerging markets such as Southeast Asia and Latin America, the primary demand is for primary hip replacement kits that offer reliability and ease of use for general orthopedic surgeons.
Local Adaptability: Our acetabular cups are designed with multiple sizing options to cater to various ethnic bone structures across the globe.
Veterinary Applications: We have expanded our precision engineering into the veterinary field, providing Total Hip Replacement (THR) kits for canines, a booming sector in the pet care industry.
Our Total Hip Replacement Metal Titanium Implants represent the pinnacle of Chinese manufacturing excellence. Titanium’s modulus of elasticity is closer to human bone than any other metal, minimizing "stress shielding" and promoting long-term bone health around the acetabular cup.

Chengdu Medev Medical Instruments Co., Ltd. stands as a beacon of high-end medical manufacturing in China. For over two decades, we have bridged the gap between premium quality and competitive pricing. Our 18,000 ㎡ facility is equipped with the latest CNC technology and aseptic workshops, ensuring every acetabular cup meets rigorous international standards.
Utilizing Hardness Testers and Ultrasonic cleaning to ensure every implant is sterile and structurally sound before shipment.
Our team of engineers focuses on continuous innovation, refining designs based on feedback from top orthopedic surgeons worldwide.
With a registered capital of 20 million Yuan and a vast production area, we handle bulk orders for national tenders with ease.
Chengdu Medev Medical Instruments Co., Ltd., Ltd was founded in 2001, covers an area of 18,000 ㎡ , including a floor area of over 15,000 ㎡ . Its registered capital reaches 20 million Yuan. which is specialized in product research, design and manufacturing of orthopedic implant instruments. As the leader in orthopedic implants and instruments manufacturing, We have been successfully supplying to 1000+ clients in 120+ countries for over 20 years thanks to extensive know-how and expertise. We adhere to the ‘people-oriented, integrity first, continuous innovation, the pursuit of excellence,’ the concept of guarding human health!

Cleaned implants have a cleaner surface, helping to reduce the risk of post-operative infection. Ensures that the implant is safer to apply to the patient during surgery.

Tests the tensile strength, yield strength and elongation of materials. Simulates the tensile forces to which the implant is subjected in the body.

Packaging should ensure a good seal to prevent the entry of external contaminants. Use heat sealing, pressing or gluing to ensure integrity.

Our production base in Chengdu represents the forefront of Chinese orthopedic implant technology and distribution capability.