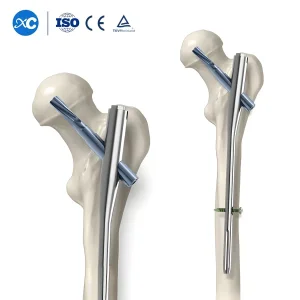
 Trauma Surgery Implants Gama Interlocking Pfna Intramedullary Nail
Trauma Surgery Implants Gama Interlocking Pfna Intramedullary Nail
 Variable Angle Distal Volar Radius Compression LCP Orthopaedic Trauma Surgery Locking Fixation
Variable Angle Distal Volar Radius Compression LCP Orthopaedic Trauma Surgery Locking Fixation
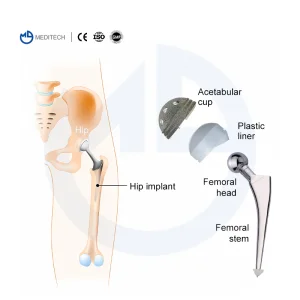 Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement
Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement
 Maxillofacial Plastic Cosmetic Surgery Micro Straightbridge Plate Trauma Series
Maxillofacial Plastic Cosmetic Surgery Micro Straightbridge Plate Trauma Series
 Plastic Cosmetic Surgery X-Shaped Plate Orthopedic Maxillofacial Implant
Plastic Cosmetic Surgery X-Shaped Plate Orthopedic Maxillofacial Implant
 Maxillofacial Micro Arc Plate 6 Holes Trauma Implant Series Plastic Cosmetic Surgery
Maxillofacial Micro Arc Plate 6 Holes Trauma Implant Series Plastic Cosmetic Surgery
 Maxillofacial Micro Straight Plate 1.0 Trauma Implant 8 Holes Plastic Cosmetic Surgery
Maxillofacial Micro Straight Plate 1.0 Trauma Implant 8 Holes Plastic Cosmetic Surgery
 Maxillofacial Micro T-Plate 7 Holes Trauma Implant Series Orthopedic Surgery
Maxillofacial Micro T-Plate 7 Holes Trauma Implant Series Orthopedic Surgery
The global orthognathic surgery market is witnessing an unprecedented transformation, driven by technological advancements and an increasing emphasis on both functional and aesthetic outcomes. As of 2024, the demand for maxillofacial implants and orthognathic correction devices has surged, particularly in North America, Europe, and the rapidly developing Asia-Pacific region. This growth is not merely clinical; it is industrial. Hospitals and specialized craniofacial centers are seeking more durable, biocompatible, and precision-fitted solutions to minimize patient recovery time and improve surgical success rates.
In the commercial sector, the shift from standardized "off-the-shelf" products to bespoke OEM/ODM manufacturing is the most significant trend. Distributors and healthcare providers now prioritize partners who can provide customized titanium plates and screws that cater to specific ethnic anatomical variations—a process known as anatomical localization. This has placed Chinese manufacturers at the forefront of the global supply chain, offering high-tech manufacturing at scale without compromising on international quality standards.
The maxillofacial implant market is projected to reach billions in USD by 2030, with a CAGR exceeding 6% annually.
Titanium Grade 4 and Grade 5 remain the gold standard, offering superior osseointegration and strength.
Cross-border medical device trade relies on stringent certifications like ISO 13485 and CE marking.
The industry is moving toward "Digital Orthognathics." The integration of AI-driven surgical planning and 3D modeling is revolutionizing how implants are manufactured. Modern factories are no longer just casting metal; they are using CAD/CAM technologies to create micro-plates and screws that match the patient's unique bone structure with sub-millimeter precision.
Another rising trend is the use of surface-treated implants. Technologies such as ultrasonic cleaning and specialized anodic oxidation improve the fatigue resistance of the implants, ensuring they can withstand the dynamic forces of the human jaw. As an OEM/ODM leader, we stay ahead by adopting these technologies early, ensuring our trauma and plastic surgery series—including micro-straight and arc plates—meet the futuristic needs of robotic-assisted surgeries.

Reduces post-operative infection risks via sterile surface preparation.

Ensures tensile strength and yield stability for high-stress implants.
Chengdu Medev Medical Instruments Co., Ltd., founded in 2001, operates from a massive 18,000 ㎡ facility with a registered capital of 20 million Yuan. We are specialized in the research, design, and manufacturing of orthopedic and orthognathic implant instruments. For over two decades, we have adhered to the concept of "guarding human health" through excellence in engineering.
Continuous innovation in maxillofacial and spinal fixation systems, catering to trauma and cosmetic surgery needs.
High-capacity OEM/ODM lines for global distributors, ensuring stable lead times and competitive factory pricing.
Aseptic workshop packaging ensures seal integrity through heat sealing and strict contaminant control.
Why do global enterprises choose Chinese factories for their orthognathic surgery implants? The answer lies in the balance between technological agility and cost-efficiency. Modern hospitals in the Middle East, Southeast Asia, and Latin America require implants that can be adapted to local surgical protocols. For example, our micro-maxillofacial plates are designed to be extremely thin yet high-strength, making them ideal for delicate cosmetic reconstructive surgeries where scarring must be minimized.
Localization is not just about the product; it's about the service. Our OEM/ODM services allow clients to request specific hole configurations, plate curvatures (like our Micro Arc Plate series), and specialized instrument sets (ACL/PCL surgery sets). By understanding the nuances of different medical regulatory environments, we ensure that every export batch is ready for immediate clinical application, backed by comprehensive testing data including hardness, elongation, and tensile strength simulation.

Inside our ISO-Certified Aseptic Packaging Workshop
 Orthopedic Surgical Medical Acl Surgery Arthroscopy Instrument Set
Orthopedic Surgical Medical Acl Surgery Arthroscopy Instrument Set
 Spine Surgical Pedicle Screw Titanium Surgery Orthopedic Implants
Spine Surgical Pedicle Screw Titanium Surgery Orthopedic Implants
 Skull Fracture Surgery Titanium Surgical Bone Locking Screw Cranial Plate
Skull Fracture Surgery Titanium Surgical Bone Locking Screw Cranial Plate
 Fracture Surgery Titanium Distal Radius Locking Plate Orthopedic Implant
Fracture Surgery Titanium Distal Radius Locking Plate Orthopedic Implant
 Orthopedic Surgery Trauma Manufacturer Finger External Fixator
Orthopedic Surgery Trauma Manufacturer Finger External Fixator
 Acl Pcl Surgery Instrument Orthopedic Metal Arthroscopy Suture Anchors
Acl Pcl Surgery Instrument Orthopedic Metal Arthroscopy Suture Anchors
 Titanium Maxillofacial Orthopedic Set of Plate Surgical Instrument Set
Titanium Maxillofacial Orthopedic Set of Plate Surgical Instrument Set
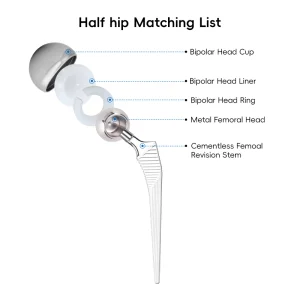 Orthopedic Tkr/Thr Surgery Hip and Knee Prosthesis Total Knee replacement
Orthopedic Tkr/Thr Surgery Hip and Knee Prosthesis Total Knee replacement
As a leading Chinese factory, we invest heavily in the final stages of the production cycle to ensure patient safety. Our Ultrasonic Cleaning process removes microscopic debris and contaminants from the implant surface, which is critical in preventing chronic inflammation or infection. This is followed by rigorous testing using our Hardness Tester, which verifies that every titanium alloy batch can handle the mechanical loads of mastication and movement.
Finally, our Aseptic Workshop Packaging utilizes state-of-the-art heat-sealing technology. This ensures a 100% sterile barrier until the product reaches the operating theater. This end-to-end control—from raw titanium to sterile package—is why Chengdu Medev Medical is the preferred OEM partner for orthopedic surgeons globally.
