Unlocking the Future of Spinal Care: China's Role in Interbody Fusion Technology
In the landscape of modern spinal surgery, Interbody Fusion Cages have evolved from simple stabilizing spacers to highly engineered, bioactive implants. As a leading manufacturer and supplier, Chengdu Medev Medical Instruments Co., Ltd. sits at the intersection of precision engineering and clinical necessity. We understand that surgeons and hospitals are not just looking for hardware; they are looking for reliable, high-performance solutions that improve patient outcomes.
Why China? The Supply Chain Resilience Factor
The perception of Chinese medical manufacturing has undergone a radical transformation. Today, it is synonymous with "High-Precision Efficiency." By integrating ISO 13485 standards, automated CNC machining, and rigorous sterilization protocols (like our in-house Ultrasonic Cleaning and Aseptic Packaging), we provide the same quality standard as Western competitors but with the distinct agility of the Chinese supply chain. This resilience ensures that even in volatile global markets, hospitals receive critical orthopedic implants without the bottleneck delays.
Technology Roadmap: The Next Generation of Fusion Cages
The future of spinal fusion lies in additive manufacturing (3D printing) and material science innovation. Our roadmap focuses on:
- Osteointegration Optimization: Moving beyond solid titanium. We are advancing porous titanium structures that mimic the trabecular bone's modulus of elasticity, significantly reducing the risk of "stress shielding."
- PEEK vs. Titanium Hybridization: Combining the radiolucency of PEEK with the bioactivity of titanium coatings, giving surgeons the best of both worlds—clear visibility for post-op assessment and superior bone-implant bonding.
- Biomechanical Stability: Utilizing Finite Element Analysis (FEA) in our R&D to simulate human spinal loads, ensuring that every cage we manufacture offers optimal subsidence resistance and sagittal balance restoration.
Localized Applications & Clinical Scenarios
Interbody fusion is not a "one size fits all" procedure. Our expertise spans:
- Cervical Fusion (ACDF): Designing low-profile, smooth-contoured cages to minimize dysphasia risks.
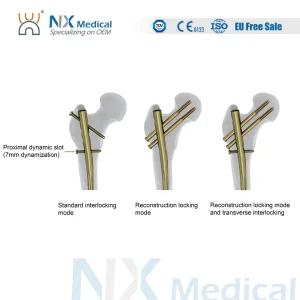
- Lumbar Fusion (TLIF/PLIF/LLIF): Tailoring cage geometry to accommodate various surgical approaches, ensuring ease of insertion and optimal lordotic correction.
We work closely with clinical partners to understand localized surgical preferences, ensuring our instruments align with the specific dexterity requirements of surgeons worldwide.
Global Industrial Status & Compliance
In the global medical devices market, trust is the currency. Since 2001, we have operated with a "People-Oriented, Integrity First" philosophy. We are not just a supplier; we are a strategic partner. Our compliance framework covers international regulatory requirements, ensuring that every product—from our locking plates to our specialized cages—meets the stringent safety benchmarks required for clinical implantation.
Frequently Asked Questions (FAQ)
Q: Why choose Chinese manufacturers for spinal implants?
A: Efficiency and innovation. China has invested heavily in high-end medical manufacturing, utilizing advanced materials and automated quality control, allowing for cost-effective pricing without sacrificing clinical safety.
Q: How do you ensure the quality of Interbody Fusion Cages?
A: We employ a multi-stage testing protocol. From hardness testing and material analysis to ultrasonic cleaning and aseptic packaging, every unit is verified against international medical device standards before leaving our facility.
Q: Can you provide OEM/ODM services?
A: Yes, as a leader in orthopedic instruments, we provide full-scale OEM/ODM services, supporting our partners from initial design concept to market-ready product manufacturing.
 Medev Medical
Medev Medical