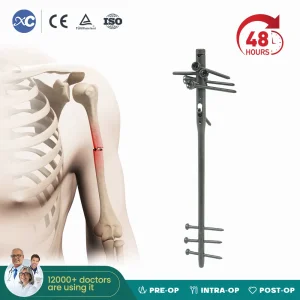

Canwell Tibial Nail Intramedullary Nail Interlocking Cannualted Nail Canetn Gamma Nail


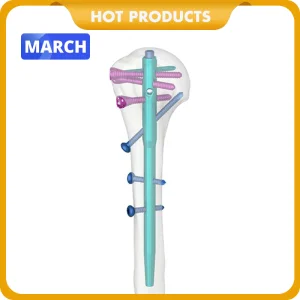

China Factory Nx Medical Orthopedic Trauma Implant Titanium Retrograde Tibial Intramedullary Nail
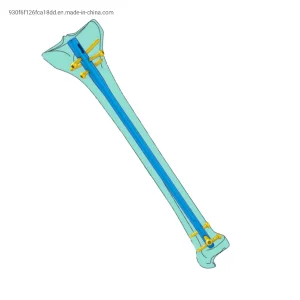
Intramedullary Nail System Expert Tibial Interlocking Nail Suprapatellar Approach

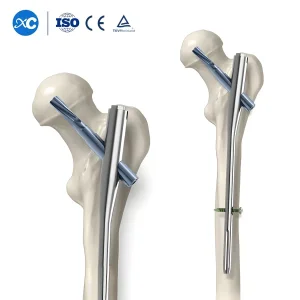
Orthopedic Surgical Implant Titanium Pfna Proximal Femoral Intramedullary Nail
The global orthopedic trauma market is witnessing a paradigm shift, with Tibial Intramedullary Nails emerging as the "Gold Standard" for long bone fracture fixation. As populations age in developed nations and industrialization increases road traffic incidents in emerging economies, the demand for high-strength, biocompatible intramedullary systems has reached an all-time high. Modern medical engineering now demands more than just a metal rod; it requires intelligent designs that facilitate minimally invasive surgery, early weight-bearing, and accelerated bone healing.
Today, the industry is valued at billions of dollars, with a compound annual growth rate (CAGR) exceeding 6%. Key markets like the United States, Germany, and Japan focus on premium-tier specialized nails, while the Asia-Pacific and Latin American regions are driving massive volume growth. China, as a global manufacturing powerhouse, has transitioned from a basic producer to a center of innovation, offering CE and FDA-compliant orthopedic solutions that rival the quality of traditional Western brands at significantly more competitive price points.
The technical evolution of Tibial Intramedullary Nails is currently focused on three major pillars: Material Science, Geometry Optimization, and Surgical Approach.
Demand high-turnover inventory with universal instrument sets that allow surgeons to handle various fracture patterns (proximal, shaft, or distal) with a single system.
Seek reliable OEM/ODM partners in China who can provide consistent quality, localized regulatory documentation, and customizable branding options.
Focus on cost-effectiveness and high-volume supply capabilities. Chinese manufacturers like Chengdu Medev are top contenders for large-scale public health contracts.
Our Tibial Intramedullary Nails are engineered for diverse clinical environments across the globe:
Scenario A: High-Altitude Trauma (Regional Clinics)
In mountainous regions where traffic accidents are frequent, our nails' interlocking design provides immediate stability, allowing patients to be transported safely after stabilization.
Scenario B: Geriatric Fracture Management (Developed Urban Centers)
With the rise of osteoporosis-related fractures, our nails feature specialized proximal locking options to provide better "purchase" in low-density bone, preventing implant migration.
Scenario C: Battlefield & Disaster Relief
The portability and simplified instrumentation of our cannulated nail systems make them ideal for rapid deployment in mobile surgical units during humanitarian crises.
The "Made in China" label in the medical device sector now stands for Advanced Innovation and Efficiency. Leading factories like Chengdu Medev Medical have invested heavily in Swiss-imported CNC machines and German testing equipment to ensure every Tibial Nail meets micron-level tolerances.
By choosing a top China manufacturer, global buyers benefit from an integrated supply chain. From raw titanium smelting to final sterile packaging, the entire process is localized, reducing lead times from months to weeks. Furthermore, our R&D teams collaborate with top-tier orthopedic surgeons in Asia to refine designs, ensuring they are ergonomic and anatomically precise for a wide range of patient ethnicities.
Strict adherence to ISO 13485 and international standards ensures our products are ready for registration in any global market.
We can prototype and refine new orthopedic instruments faster than anywhere else, allowing our partners to stay ahead of market trends.
Whether it's a small order for a private clinic or a 40ft container for a national healthcare system, we have the logistical infrastructure to deliver.
Chengdu Medev Medical Instruments Co., Ltd., Ltd was founded in 2001, covers an area of 18,000 ㎡ , including a floor area of over 15,000 ㎡ . Its registered capital reaches 20 million Yuan. which is specialized in product research, design and manufacturing of orthopedic implant instruments. As the leader in orthopedic implants and instruments manufacturing, We have been successfully supplying to 1000+ clients in 120+ countries for over 20 years thanks to extensive know-how and expertise. We adhere to the ‘people-oriented, integrity first, continuous innovation, the pursuit of excellence,’ the concept of guarding human health!

Cleaned implants have a cleaner surface, helping to reduce the risk of post-operative infection. Ensures that the implant is safer to apply to the patient during surgery.

Tests the tensile strength, yield strength and elongation of materials. Simulates the tensile forces to which the implant is subjected in the body.

Packaging should ensure a good seal to prevent the entry of external contaminants. Use heat sealing, pressing or gluing to ensure the integrity of the package.

Our 18,000 ㎡ state-of-the-art facility ensures every orthopedic instrument meets the highest global standards of precision.
Trauma Surgery Implants Gama Interlocking Pfna Intramedullary Nail
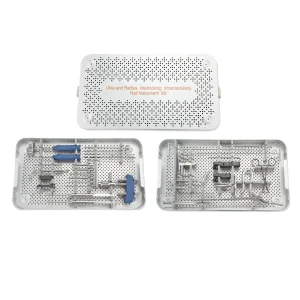
Orthopedic Medical Device Surgical Ulna and Radius Interlocking Intramedullary Nail Instrument Set

High-Quality Interlocking Nail Intramedullary Nail in Blue Green Gold Colors
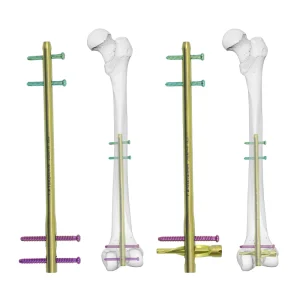
Medical Supply Orthopedic Implant Titanium Dfn Distal Femoral Intramedullary Nail
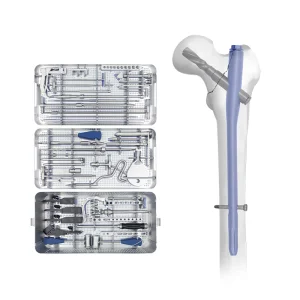
Medical Supply Orthopedic Implant Titanium Femur Pfna Intramedullary Nail Instrument Set

Orthopedic Implant Interlocking Nail Standard Intertan Intramedullary Nail for Fracture Surgery


Intramedullary Nail System Expert Humeral Proximal Interlocking Nail Blade Locking