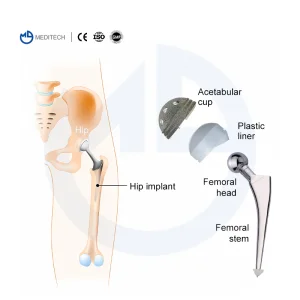
 Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement
Hip Artificial Knee Joint Orthopedic Implants Cemented Stem Hip Replacement S-ROM Titanium Alloy Cementless Hip Joints for Revision Arthroplasty Surgery
S-ROM Titanium Alloy Cementless Hip Joints for Revision Arthroplasty Surgery Total Hip Replacement Prosthesis Corail Femoral Stem, Artificial Hip Joint System Titanium Hip Joint Prosthesis Implant
Total Hip Replacement Prosthesis Corail Femoral Stem, Artificial Hip Joint System Titanium Hip Joint Prosthesis Implant Primary Total Hip System Hip Joint Replacement Surgery Femoral Head
Primary Total Hip System Hip Joint Replacement Surgery Femoral Head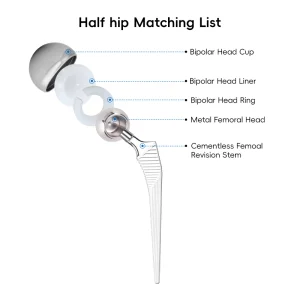 Orthopedic Tkr/Thr Surgery Hip and Knee Prosthesis Total Knee Joint Replacement Instrument Set
Orthopedic Tkr/Thr Surgery Hip and Knee Prosthesis Total Knee Joint Replacement Instrument Set Total Hip and Knee Joint Replacement Metal Titanium Implant Made in China
Total Hip and Knee Joint Replacement Metal Titanium Implant Made in China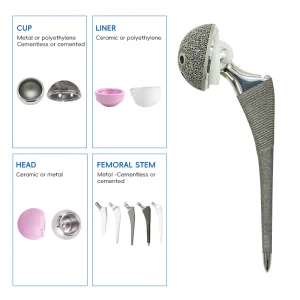 UHMWPE Insert Acetabular Cup Hip Joint Hemiarthroplasty System Total Knee Replacement Prosthesis Implants Instruments Set
UHMWPE Insert Acetabular Cup Hip Joint Hemiarthroplasty System Total Knee Replacement Prosthesis Implants Instruments Set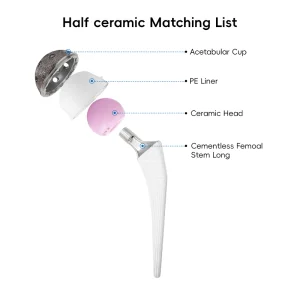 Orthopedic Prosthesis Implant Artificial Hip and Knee Joint Replacement
Orthopedic Prosthesis Implant Artificial Hip and Knee Joint ReplacementThe global orthopedic market is witnessing a significant shift towards revision surgeries. Revision hip replacement, a procedure performed when a primary hip implant fails due to aseptic loosening, infection, or periprosthetic fractures, has become a multi-billion dollar sector. As life expectancy increases globally, patients are outliving their primary implants, necessitating advanced revision hip components that can handle bone loss and compromised tissue.
Industrial Fact: The Global Revision Hip Replacement Market is projected to grow at a CAGR of over 5.8% through 2030, driven by the aging demographic in North America, Europe, and Asia-Pacific.
Revision surgeries are inherently more complex than primary ones. Manufacturers are now focusing on "Modular Systems" that allow surgeons to customize the implant size and geometry intraoperatively to match the specific anatomy and bone deficiency of the patient. This modularity is the cornerstone of modern revision hip arthroplasty.
Additive manufacturing (3D printing) is revolutionizing how we create porous surfaces. These structures mimic trabecular bone, promoting superior biological fixation in revision cases where bone quality is poor.
To combat the high risk of dislocation in revision surgeries, dual-mobility acetabular components are becoming a standard choice, offering increased range of motion and stability.
Pre-operative digital templating using AI allows surgeons to predict the necessary component sizes and configurations for complex revision cases, reducing surgery time.
Hospitals and surgical centers worldwide are seeking reliable supply chains for revision hip components. The demand isn't just for the implants themselves but for complete instrument sets that ensure surgical precision. Procurement officers prioritize:
As a leading hub for medical device manufacturing, China offers a unique combination of technological prowess and cost-efficiency. Our facilities represent the pinnacle of this industrial evolution.
China's "Medical Valley" regions have developed comprehensive supply chains, from raw material sourcing (high-grade titanium and UHMWPE) to advanced surface treatment technologies like HA (Hydroxyapatite) coating and plasma spraying. This allow us to provide Revision Hip Components that are 30-50% more cost-effective than Western counterparts without compromising on clinical safety.
Chengdu Medev Medical Instruments Co., Ltd., Ltd was founded in 2001, covers an area of 18,000 ㎡ , including a floor area of over 15,000 ㎡ . Its registered capital reaches 20 million Yuan. which is specialized in product research, design and manufacturing of orthopedic implant instruments. As the leader in orthopedic implants and instruments manufacturing, We have been successfully supplying to 1000+ clients in 120+ countries for over 20 years thanks to extensive know-how and expertise. We adhere to the ‘people-oriented, integrity first, continuous innovation, the pursuit of excellence,’ the concept of guarding human health!

Cleaned implants have a cleaner surface, helping to reduce the risk of post-operative infection. Ensures that the implant is safer to apply to the patient during surgery.

Tests the tensile strength, yield strength and elongation of materials. Simulates the tensile forces to which the implant is subjected in the body.

Packaging should ensure a good seal to prevent the entry of external contaminants. Use heat sealing, pressing or gluing to ensure the integrity of the package.

Our facility utilizes 5-axis CNC machining centers to ensure the complex geometry of revision stems and cups meets sub-micron tolerances.
Revision hip components from Chengdu Medev are designed for the most challenging clinical scenarios, including:
Our components, such as the S-ROM titanium alloy systems, are engineered to provide anatomical restoration of the hip's center of rotation, ensuring that patients regain their mobility and quality of life with long-term implant survival rates.
We invest 15% of our annual revenue into R&D, collaborating with orthopedic surgeons to refine the ergonomics of our instrument sets. This ensures that when a surgeon uses a Chengdu Medev kit, the process is intuitive, fast, and precise.
 Manufacturer Rsk Femoral Condyle Revision Tka Total Knee Artificial Replacement
Manufacturer Rsk Femoral Condyle Revision Tka Total Knee Artificial Replacement Manual Hip Joint Femoral Instrument Set Standard Stainless Instrument Total Knee Joint Replacement Instrument Set Hip and Knee
Manual Hip Joint Femoral Instrument Set Standard Stainless Instrument Total Knee Joint Replacement Instrument Set Hip and Knee Wholesale High Quality Hip Joint Femoral Instrument Set Bone Reduction Total Knee Joint Replacement Instrument Set Hip and Knee
Wholesale High Quality Hip Joint Femoral Instrument Set Bone Reduction Total Knee Joint Replacement Instrument Set Hip and Knee China Factory OEM Support Thr/Tkr Total Knee and Hip Joint Replacement Instrument Set
China Factory OEM Support Thr/Tkr Total Knee and Hip Joint Replacement Instrument Set Orthopaedic Ti-Alloy Cementless Thr Total Hip Joint Replacement
Orthopaedic Ti-Alloy Cementless Thr Total Hip Joint Replacement Best Performance Surgical Power Drill Total Hip Replacement
Best Performance Surgical Power Drill Total Hip Replacement Total Hip Replacement for Osteoarthritis Secondary to Hip Dysplasia
Total Hip Replacement for Osteoarthritis Secondary to Hip Dysplasia Advanced Hip & Knee Prosthesis with Durable Materials
Advanced Hip & Knee Prosthesis with Durable Materials