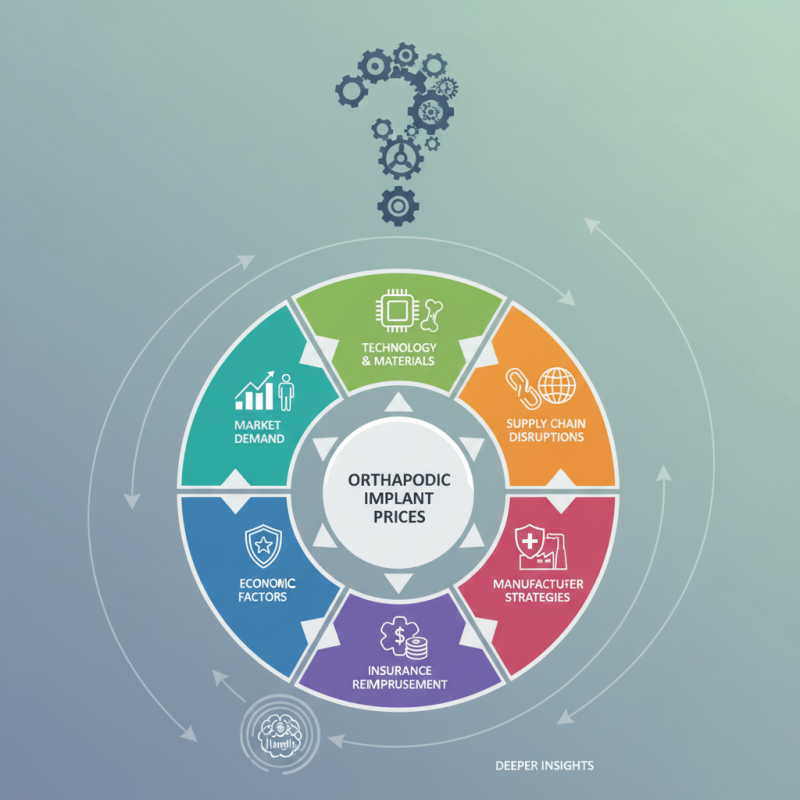
The healthcare industry constantly faces varying prices for medical devices, particularly orthopedic implants. As hospitals and patients navigate these costs, a common question arises: "Why do orthopedic implants prices fluctuate?" The answers are complex, interwoven with multiple factors. Market demand plays a vital role, as an increase in surgeries directly influences prices. Additionally, advancements in technology and materials can cause significant changes in production expenses.
Supply chain disruptions also contribute to price variability. For example, pandemic-related shortages have led to unexpected price hikes. Manufacturers may have different pricing strategies based on quality and brand reputation, further complicating the landscape. Understanding these dynamics is essential for consumers and healthcare providers alike.
Moreover, insurance reimbursement policies can create additional price fluctuations. Some implants may be covered better than others, impacting patient costs directly. Reflecting on these elements helps stakeholders better navigate the unpredictable nature of implant pricing, revealing deeper insights into market behaviors and consumer impacts.
The prices of orthopedic implants can vary widely due to several critical factors. Supply chain issues contribute significantly, impacting costs. A 2022 report from the Orthopedic Industry Association highlighted that over 30% of all medical device manufacturers cited supply chain disruptions as a primary factor for price changes. Inflation and shipping costs lead to inconsistent pricing across regions, creating disparities that can confuse those seeking treatment.
Regulatory standards also play an essential role in pricing. Different countries have varying regulations affecting how implants are developed and priced. For instance, the average cost of a knee implant in the United States can range from $10,000 to $50,000, influenced by local healthcare policies and reimbursement rates. Comparatively, systems in Europe often showcase more uniform pricing, driven by stricter regulatory oversight and negotiated purchasing agreements.
Market demand leads to fluctuations as well. An increasing aging population fuels the need for joint replacements, further driving up prices. Interestingly, hospitals may inflate costs due to their own financial challenges, leading to higher patient expenses. Overall, these factors create a complex landscape where pricing is ever-changing and often unpredictable.
The chart below illustrates the fluctuations in prices of various orthopedic implants over the last five years, highlighting key factors that influence these changes.
The quality of materials used in orthopedic implants significantly impacts their pricing. High-grade titanium alloys, for instance, are durable and biocompatible. This quality justifies higher costs. Conversely, cheaper materials may reduce expenses but can lead to complications post-surgery. Patients must weigh the benefits of lower prices against potential risks.
Manufacturers often face a dilemma. They can cut costs by using lower-end materials, or they can invest in research for better quality. Research indicates that the long-term health of patients is more favorable with higher-quality implants. However, not all patients prioritize long-term outcomes when considering price. This discrepancy creates a dynamic market, leading to fluctuating prices.
Moreover, supply chain issues can also affect implant prices. Material shortages or increased demand can push prices up unexpectedly. It’s essential for stakeholders to understand these nuances. Transparency about materials and pricing can help patients make informed decisions. So, while lower prices are appealing, they may not always reflect the best choice for health and recovery.
The landscape of orthopedic implants is shaped by extensive research and development. Innovations in materials and techniques can dramatically alter production costs. Each advancement has the potential to improve patient outcomes but often comes with substantial financial investments. Companies must weigh the balance between cutting-edge technology and cost efficiency.
Research drives variability in prices. Every study requires funding, time, and expertise. As new materials are tested, companies explore various methods to enhance durability and compatibility. Some projects may yield promising results, while others may not meet expectations. This unpredictability contributes to fluctuations in pricing.
Quality assurance also plays a crucial role. Rigorous testing ensures safety and effectiveness. The cost associated with compliance to regulatory standards impacts final pricing. This can lead to higher prices for products that undergo stringent evaluations. Finding the right balance between affordability and innovation is an ongoing challenge in the industry.
The market for orthopedic devices is influenced by various factors. Demand for these products fluctuates based on demographic changes, advancements in medical technology, and public health concerns. Aging populations often require more orthopedic procedures. This results in increased demand for related implants.
Supply trends can also impact pricing. Manufacturers face challenges in production costs, regulatory compliance, and material availability. Raw material prices can rise unexpectedly, leading to higher production costs. Additionally, tariffs and trade policies may contribute to supply chain disruptions.
Market competition affects prices significantly. New players enter the field, attempting to meet the growing demand. However, some companies struggle with scaling their production. This imbalance can lead to price instability. Stakeholders must carefully monitor both supply and demand dynamics to make informed decisions. Understanding these trends helps navigate the complex landscape of orthopedic device pricing.
Regulatory influences play a significant role in the pricing strategies for orthopedic implants. Regulatory bodies enforce strict guidelines that impact production costs and market accessibility. For instance, in the United States, the FDA requires rigorous testing before implants can be marketed. This process can take years and may be costly for manufacturers. According to a report by MedView, regulatory compliance can add up to 25% to the overall cost of an implant.
The complexity of global regulations further complicates pricing. Different countries have varying standards and approval processes, which may lead to price discrepancies for the same product. In the European Union, the Medical Device Regulation (MDR) requires additional documentation and oversight. This can delay market entry and drive up prices. A report from GlobalData indicates that regions with more stringent regulations often see a 15% to 30% increase in implant prices compared to markets with less oversight.
Moreover, the impact of reimbursement policies cannot be overlooked. Insurance coverage and payment models can dictate the pricing strategies used by manufacturers. According to a 2022 study by the American Journal of Orthopedics, successful reimbursement strategies often hinge on demonstrating the clinical efficacy of implants. This ongoing need for evidence adds layers of cost and complexity to pricing, often leading to further fluctuations.
| Implant Type | Average Price ($) | Price Range ($) | Regulatory Influence |
|---|---|---|---|
| Total Hip Replacement | 25,000 | 20,000 - 30,000 | FDA Approval Processes |
| Knee Implant | 15,000 | 10,000 - 20,000 | Post-Market Surveillance Regulations |
| Spinal Fusion Device | 30,000 | 25,000 - 35,000 | Clinical Trial Guidelines |
| Shoulder Implant | 22,000 | 18,000 - 26,000 | International Standards Compliance |
| Ankle Implant | 18,000 | 15,000 - 22,000 | Regulatory Pathway Variability |
: The quality of materials used significantly impacts pricing. High-grade materials justify higher costs.
Cheaper materials may lower costs but can lead to complications post-surgery.
Not all patients consider long-term benefits when deciding on price.
Material shortages or increased demand can unexpectedly raise prices.
Innovations can alter production costs, improving outcomes but requiring financial investment.
Variability in research funding and project outcomes contributes to unpredictable pricing.
Compliance with safety standards can lead to higher prices for rigorously tested products.
While appealing, lower prices may not reflect the best choice for health and recovery.
Finding this balance remains a challenge in the industry due to costs and patient needs.
Patients must weigh material quality against potential risks and long-term health outcomes.
The article "Why Do Orthopedic Implants Prices Fluctuate: Best Insights?" delves into the various factors influencing the pricing of orthopedic implants. It highlights that the quality of materials used in manufacturing plays a significant role in determining costs, with advanced materials often leading to higher prices. Additionally, the article discusses how research and development investments contribute to cost variability, as ongoing innovations can result in more effective yet pricier implants.
Moreover, market demand and supply trends substantially impact pricing strategies, with changes in healthcare policies and consumer needs shaping the dynamics of the orthopedic device market. Regulatory factors also come into play, as compliance with safety standards can lead to varying costs for manufacturers. Overall, understanding these influences is essential to answer the question, "Why do orthopedic implants prices fluctuate?" as it reveals a complex interplay of material quality, innovation, market demand, and regulations.

 Medev Medical
Medev Medical