Chengdu Medev Medical Instruments Co., Ltd. was founded in 2001, covers an area of 18,000 ㎡, including a floor area of over 15,000 ㎡. Its registered capital reaches 20 million Yuan. We are specialized in product research, design, and manufacturing of high-end orthopedic implant instruments. As a premier Anterior Cervical Plating Systems Exporter, we have successfully supplied to 1000+ clients in 120+ countries for over 20 years.
Our commitment to excellence is reflected in our adherence to the "people-oriented, integrity first, continuous innovation, and pursuit of excellence" philosophy. We are not just a manufacturer; we are a partner in guarding human health through advanced spinal fixation technologies.

The global market for Anterior Cervical Plating Systems is experiencing unprecedented growth, driven by an aging population and the increasing prevalence of degenerative disc diseases. As a leading exporter, we observe that hospitals and surgical centers are shifting toward minimally invasive procedures that require low-profile, high-stability implants.
The move towards titanium alloy (Grade 5) and bio-compatible materials ensures better osseointegration and reduced imaging artifacts during post-operative MRI/CT scans.
Integration of robotic-assisted surgery and 3D-guided navigation is necessitating more precise and standardized cervical plating systems for perfect fitment.
Global procurement teams are prioritizing CE and ISO-certified suppliers who can offer a balance between surgical reliability and cost-effectiveness.
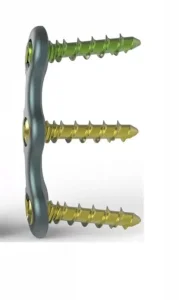
Our Anterior Cervical Fixation System represents the pinnacle of orthopedic engineering. Designed for patients suffering from cervical disc herniation or trauma, these plates provide rigid stability while maintaining a low profile to minimize esophageal irritation.
Click below to explore our CE-certified range:
View Technical SpecificationsAs a leading Anterior Cervical Plating Systems Exporter based in China, Chengdu Medev leverages advanced manufacturing clusters and supply chain efficiencies to deliver world-class medical devices. Our competitive edge lies in the combination of high-precision CNC machining and rigorous quality control protocols.
Our systems are designed for diverse clinical environments, from high-traffic emergency trauma centers in North America to specialized spinal clinics in the Middle East. Whether it's a single-level ACDF (Anterior Cervical Discectomy and Fusion) or multi-level complex reconstruction, our plating systems provide the versatility surgeons demand.

Quality is the cornerstone of our operations. Every implant undergoes a series of stringent tests to ensure patient safety and surgical success.

Cleaned implants have a cleaner surface, helping to reduce the risk of post-operative infection. Ensures that the implant is safer to apply to the patient during surgery.

Tests the tensile strength, yield strength and elongation of materials. Simulates the tensile forces to which the implant is subjected in the body.

Packaging ensures a good seal to prevent contaminants. We use heat sealing and pressing in 100,000-level cleanrooms to ensure biological integrity.
The evolution of Anterior Cervical Plating Systems has transformed the outcomes of cervical spine surgeries. In the past, surgeons relied on external bracing or autografts without internal fixation, which often led to pseudoarthrosis or graft collapse. Today, as a professional exporter, we provide solutions that offer immediate biomechanical stability, allowing for early patient mobilization and higher fusion rates.
Our plates are manufactured primarily from high-grade Titanium Alloy (Ti-6Al-4V). This material is chosen for its excellent biocompatibility, high strength-to-weight ratio, and its ability to integrate with the surrounding bone. As global Anterior Cervical Plating Systems Exporters, we emphasize the importance of "Stress Shielding" prevention—our plates are designed with just enough elasticity to allow for load-sharing across the bone graft, which promotes faster healing.
Navigating the complex regulatory landscape is a challenge for many medical device distributors. Chengdu Medev simplifies this process. Our manufacturing facility is ISO 13485 certified, and our flagship products carry the CE mark. This compliance is vital for our partners in Europe, Southeast Asia, and Latin America, ensuring that every shipment meets the stringent safety standards required for human implantation.
Beyond our standard catalog, we offer comprehensive OEM and ODM services. We understand that different regions may have specific anatomical preferences or unique surgical techniques. Our R&D team works closely with clinical advisors to iterate on plate designs, screw thread pitches, and locking mechanisms to satisfy specific market demands.